
Spectro-EFC, SMA 905, 1.75 mL - Optical Fiber Spectro-Electrochemical Flow Cell
Redoxme



Price: On Request
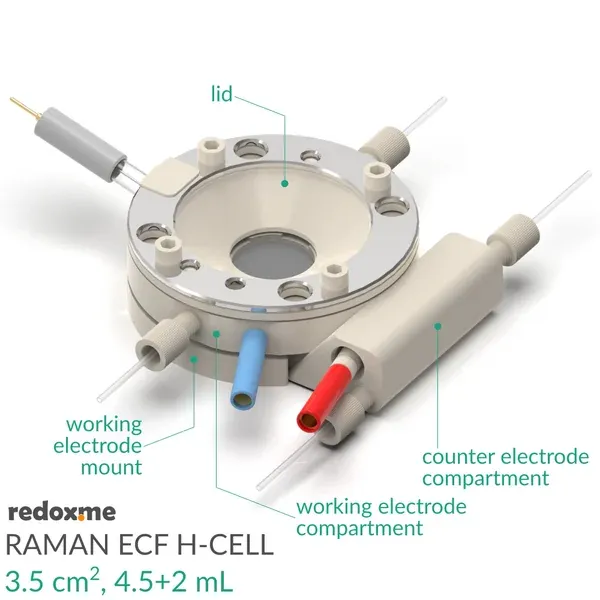
This cell combines two classical analytical techniques such as electrochemistry and Raman spectroscopy, to obtain in-situ chemical information about the reactions taking place during an electrochemical experiment. The Raman Electrochemical Flow H-Cell offers several advantages over a single-compartment Raman Electrochemical Flow Cell. The ion-exchange membrane allows for the physical separation of the anode and cathode compartments, preventing direct mixing of the reactants and products. Each compartment can have different electrolytes or pH conditions, allowing for precise control over the reaction environment. With an ion-exchange membrane, cross-talk between the anode and cathode reactions is minimized, leading to more accurate measurements of electrocatalytic activity.
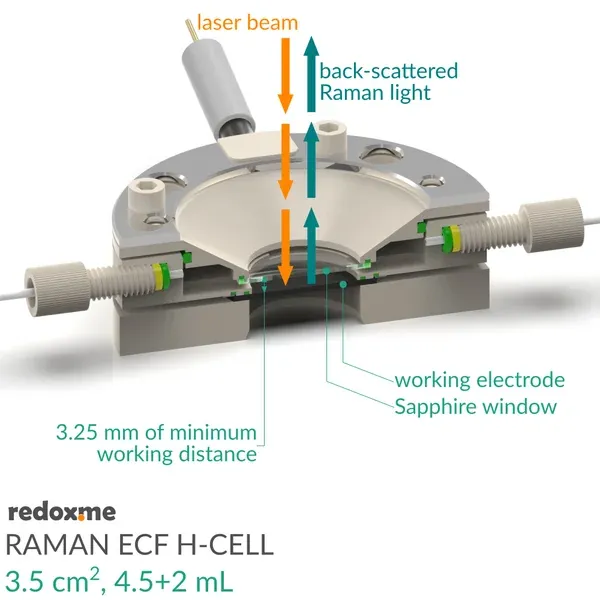
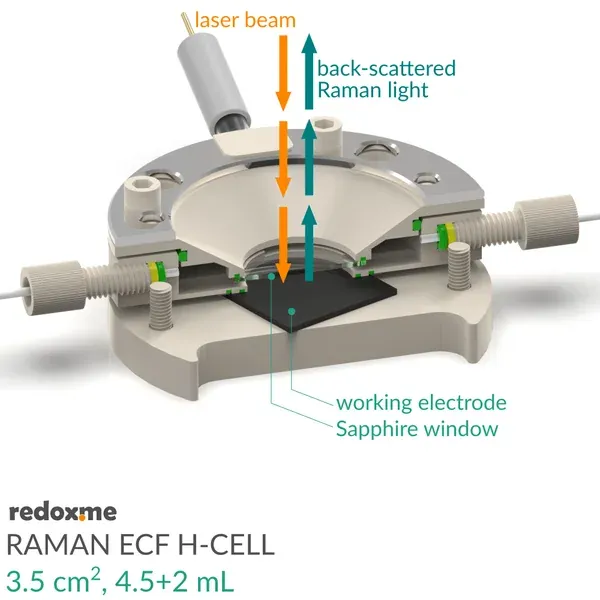
It consists mainly of four elements: (i) sample holder with Tantalum foil serving as an electric contact to working electrode surface, (ii) working electrode compartment in which reference is installed together with electrolyte inlet and outlet, (iii) lid which seals the cell and holds the Sapphire window, and (iv) counter electrode compartment which holds the ion-exchange membrane and where the counter electrode is installed. During an experiment Raman laser is focused onto the surface of a thin film working electrode through a transparent Sapphire window and thin layer of electrolyte (total optical path of 3.25 mm).
The cell elements are constructed with materials that are inert to the sample (PEEK, Fluorocarbons). It well fits aqueous (FKM O-Rings) and organic solvent (FFKM O-Rings) electrolyte requirements. The construction is gas-tight.
Application Note This cell can be used to track kinetic phenomena such as the near-surface proton concentration changes during oxidation and reduction reaction at working electrode. It can be also used to identify materials such as carbon, metal oxides, polymers and electrolytes, and to determine their structure and distribution. The use of this cell with an ion-exchange membrane provides a more controlled and reliable platform for studying electrocatalysis, allowing researchers to investigate fundamental mechanisms and optimize catalytic performance with greater precision.
Product Includes
Nominal Exposure Area
3.5 cm²
Optical Path Length
3.25 mm (incl. Sapphire window)
Electrode Plug Diameter (Cell)
6 mm
Cell Body Material
PEEK, Fluorocarbons
O-Ring Material (Aqueous Electrolyte)
FKM
O-Ring Material (Organic Electrolyte)
FFKM
Window material
Sapphire
Number of Compartments
2
Membrane Size
20 mm dia.
Spectrometer Compatibility
Raman
Electrolyte Volume
4.5 mL (WE) + 2 mL (CE)
Compatible Electrolyte Types
Aqueous and organic solvents
Sample Mount Type
Magnetic or screw mount
0 global ratings
Verified Identity
Every review undergoes an email verification process to ensure it originates from a verified industry professional.
Verified Purchase
We manually validate "Verified Purchase" claims by cross-referencing with supplier records or identifying the equipment in peer-reviewed scientific publications.
Transparent Publishing
All verified reviews, whether positive or negative, are published without bias as long as they comply with our community guidelines.
No reviews yet. Be the first to share your thoughts!
Share your experience with this product.
Help the scientific community by sharing research papers that utilize this equipment.
Contact us to add your research.