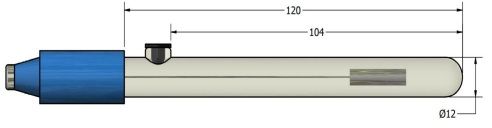
In acetonitrile, propylene carbonate, DMSO, ionic liquids and other non-aqueous electrolytes, aqueous reference electrodes leak water and shift potential. The non-aqueous Ag/Ag⁺ and Ag/AgNO₃ reference electrodes solve that: a silver wire in a controlled Ag⁺ solution, typically 0.01 M AgNO₃ in the same solvent, separated from the working compartment by a ceramic frit. This page covers Ag/AgNO₃ reference electrodes in acetonitrile and solvent-resistant single-junction formats, plus refillable silver/silver-ion bodies for organic electrochemistry, battery research and cyclic voltammetry in non-aqueous media.